The Renal System | Approach to the patient with complaints

INTRODUCTION
The patient with renal disease can present in a number of ways:
- with symptoms or physical signs that are typically associated with renal disease
- no symptoms but an abnormality is detected on clinical or laboratory examination
- with a diagnosis of systemic disease known to be associated with renal disease
- with a family history of inherited renal disease that prompts further assessment
- following exposure to nephrotoxic agents.
Cardinal symptoms suggesting the presence of underlying renal disease include disorders of micturition, disorders of urine volume, alterations in urine composition, loin pain, oedema and hypertension. In addition, a wide variety of symptoms and complications can be associated with advanced renal failure (uraemia). Renal disease in asymptomatic patients is often detected on dipstick urinalysis, biochemical analysis of the serum (and urine), and blood pressure measurement undertaken during some intercurrent illness or as part of a health screening programme.
CLINICAL HISTORY
Disorders of micturition
Frequency of micturition
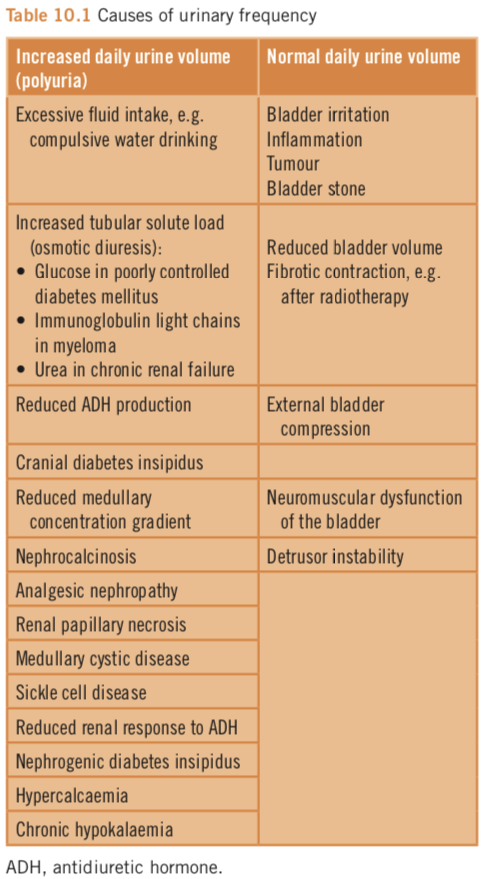
Urinary frequency is the urge to empty the bladder more often than normal. It may be associated with nocturia, urgency (a need to pass urine immediately) and incontinence. The causes of urinary frequency are listed in Table 10.1. It is important to determine whether urinary frequency is associated with normal or increased urine volumes. Normal urine volumes imply bladder dysfunction caused by inflammation, tumour or stone of the bladder, or reduced bladder capacity. Increased urine volume indicates polyuria. Nighttime frequency may also be caused by sleep disturbance. Sleep normally induces antidiuretic hormone (ADH) secretion, which causes a reduction in urine volumes. Subjects who sleep poorly have no increase in ADH secretion and therefore urine volumes remain high.
Poor urinary stream
A poor urinary stream is typically caused by bladder outflow obstruction from prostate enlargement or urethral disease. The characteristic symptoms are:
- poor urine flow with a weak stream
- difficulty in initiating micturition (hesitancy) and/or in stopping (terminal dribbling).
Urinary frequency is often also present.
Progression of the obstruction may lead to complete failure of micturition (urinary retention). Paradoxically some patients with bladder outflow obstruction produce increased urine volumes due to tubular dysfunction arising from back pressure on the kidneys. Prolonged bladder outflow obstruction can result in progressive renal dysfunction (obstructive nephropathy).
Dysuria
Pain or discomfort during micturition usually results from bladder, prostatic or urethral inflammation. It is described as a burning or tingling sensation in the urethra or suprapubic area during or immediately after micturition. When associated with frequency and urgency of micturition it indicates cystitis (bladder inflammation, usually due to bacterial infection). Perineal or rectal pain on micturition in men suggests prostatic inflammation (prostatitis).
Disorders of urine volume
Disorders of urine volume can be categorized as polyuria (an increase in urine volume), oliguria (a reduction in urine volume), and anuria (a loss of urine output).
Polyuria
This is defined as urine output over 3L per day. Patients may find it difficult to differentiate polyuria from frequency, and thirst may in fact be the presenting symptom. Measurement of the daily urine volume is therefore helpful. The causes of polyuria are listed in Table 10.1.
Oliguria
This is the reduction in urine volume below the level required for the excretion of normal metabolic byproducts. It is therefore associated with the accumulation of nitrogenous waste products (i.e. an increase in serum urea and creatinine levels) and indicates the development of acute kidney injury (AKI). Practically, oliguria can be defined as a urine volume less than 500mL/day, less than 30mL/h, or less than 0.5 mL/kg body weight/h.
Anuria
This is a urine volume of less than 100 mL/day. It is most commonly due to renal tract obstruction, but can also result from vascular catastrophes (e.g. acute renal artery occlusion), acute cortical necrosis, and inflammatory glomerular disease (e.g. Goodpasture’s disease).
Disorders of urinary composition
Haematuria
Visible haematuria is a striking symptom that usually prompts the patient to seek urgent medical attention. Haematuria from any cause can be visible or invisible, i.e. apparent only on urine dipstick testing. It can result from bleeding anywhere in the urinary tract from the glomeruli to the urethra, the commonest cause being urinary tract infection. It is important to appreciate that red/brown urine does not always indicate haematuria (see Box 10.1).
The pattern of visible haematuria gives clues to the underlying cause:
- visible haematuria present at the start of urine flow with subsequent clearing is usually due to urethral bleeding
- haematuria mainly at the end of micturition (terminal haematuria) is indicative of bladder or prostate bleeding
- glomerular haematuria is often red-brown in colour and may be described as ‘smoky’ or like coca-cola or tea
- visible haematuria that coincides with episodes of mucosal infection (often pharyngitis) or with exercise is frequently due to IgA nephropathy
- blood clots are never caused by glomerular bleeding.
Proteinuria
This is usually asymptomatic and identified by urine dipstick testing followed by more specific laboratory tests (see Investigations section). However, patients with heavy proteinuria (usually nephrotic range proteinuria (>3.5g/day)) may describe frothing of the urine on micturition.
Pneumaturia
Patients with fistulae between the urinary and alimentary tracts may describe the passage of air or ‘fizzing’ of the urine on micturition. This usually results from Crohn’s disease, diverticular disease or carcinoma of the colon.
Small pieces of tissue may be passed in the urine in patients with malignant disease of the urinary tract or papillary necrosis. In addition, solid material may enter the urinary tract from the bowel via vesicocolonic fistulae. Patients with a predisposition to stone formation may notice sand-like material in the urine.
Pain
Pain is an inconsistent feature of renal disease. When present it is usually the result of renal tract inflammation or obstruction.
- Inflammation of the kidney due to infection (acute pyelonephritis) results in pain localized to the renal angle on the affected side.
- Perirenal abscess formation may lead to diaphragmatic irritation or psoas muscle irritation depending on the direction in which the abscess tracks.
- Glomerular inflammation is usually asymptomatic. Acute and fulminant glomerulonephritis (GN) may cause a dull lumbar ache.
- The loin pain haematuria syndrome is the constellation of dull loin pain, which may be severe, and visible haematuria. It has no known cause and investigation generally reveals no structural or renal histological abnormality apart from complement (C3) deposition in the walls of afferent arterioles.
- The pain of acute renal tract obstruction is sudden in onset, severe and colicky in nature (ureteral colic). The pain often radiates to the groin, scrotum or labia.
- Chronic urinary tract obstruction in contrast is often asymptomatic. Obstruction at the level of the bladder outflow tract may lead to symptoms of hesitancy, poor flow, and terminal dribbling, but obstruction above this level may be completely asymptomatic.
Oedema
Oedema results from the accumulation of salt and water and reflects an increase in the total body sodium content. In renal disease it occurs either as part of the nephrotic syndrome or because of reduced excretory function and subsequent failure to excrete ingested salt and water. Nephrotic oedema is typically first noticed in a periorbital distribution after a period of recumbency. Facial oedema is rare in advanced renal failure, heart failure or cirrhosis because patients with those conditions are unable to lie flat due to either the development of pulmonary oedema, or pressure on the diaphragm from ascites. As the degree of oedema increases, fluid accumulates around the ankles, legs, sacrum and elbows in a gravity-dependent manner, and then becomes more generalized (Fig. 10.1). Genital oedema may ensue and cause significant distress.
A patient with facial and peri-orbital oedema is very likely to have nephrotic syndrome. This presentation almost never occurs with heart failure, cirrhosis or advanced renal failure because patients cannot lie flat due to the development of pulmonary oedema or because of splinting of the diaphragm from ascites.
Uraemia
In some patients, the underlying renal disease causes no specific symptoms and presentation only occurs once advanced renal failure has developed. The accumulation of nitrogenous waste products leads to the uraemic syndrome (Box 10.2). These symptoms are vague and rarely specific enough to identify renal failure without further investigation.
- Anorexia, nausea, vomiting, weight loss
- Restless limbs and generalized restlessness
- Fatigue and weakness
- Reduced mental acuity and mental clouding
- Loss of concentration
- Pruritus
- Peripheral dysaesthesia
- Headache
- Sleep disturbance
- Sexual dysfunction (particularly in younger patients)
- Abnormal or loss of taste
Symptoms related to systemic disease
Renal disease may be associated with a wide variety of systemic diseases (e.g. systemic lupus erythematosus, multiple myeloma) and therefore the identification of renal disease may be prompted by the development of symptoms of the underlying disease state.
THE REST OF THE HISTORY
Past medical history
Explore all health problems going back to early childhood. A large number of diseases can result in renal dysfunction either directly or as the result of complications during treatment. For example:
- a history of childhood urinary tract infections and late nocturnal enuresis may indicate the presence of vesicoureteric reflux and chronic pyelonephritis
- systemic lupus erythematosus may initially cause symptoms confined to the skin and joints, but later may progress to involve the kidneys
- other chronic inflammatory conditions such as rheumatoid arthritis or Crohn’s disease may result in AA amyloidosis with renal involvement
- chemotherapy for malignant disease with cyclophosphamide carries a long-term risk of bladder cancer which may present with haematuria.
The medical conditions associated with renal disease are summarized in Table 10.2. You should include a review of previous medical notes with particular emphasis on previous renal function tests.
Gynaecological and obstetric history
In women, seek details of the menstrual history, contraception and pregnancies.
- A delayed menarche may be caused by chronic kidney disease (CKD).
- Some women with significant renal dysfunction become amenorrhoeic, whilst others develop menorrhagia as a consequence of the associated clotting and platelet abnormalities.
- In women taking the combined oestrogen–progesterone contraceptive pill, renal disease significantly increases the risk of developing hypertension.
- Fertility is impaired in patients with renal disease.
- Renal disease may be exacerbated or initiated by pregnancy. Seek details of pregnancy-associated rises in blood pressure, the presence of proteinuria, and the duration and outcome of pregnancies.
- Although bacteriuria frequently occurs in pregnancy, upper urinary tract infection during pregnancy may indicate a structural abnormality of the upper urinary tract.
- Recurrent fetal loss may suggest the presence of the antiphospholipid antibody syndrome.
Drug history
A variety of drugs may induce or exacerbate renal disease, or may induce circulatory change that adversely affects renal perfusion.
- A single dose of a large variety of drugs may be sufficient to induce an acute allergic interstitial nephritis.
- The interstitial nephritis induced by non-steroidal anti-inflammatory agents may, in contrast, only develop after several months of treatment.
- Hypotensive drugs may cause deterioration in renal function, particularly when the blood pressure is lowered precipitously.
- Angiotensin-converting enzyme inhibitors, angiotensin-receptor blockers, and direct renin inhibitors may cause a profound loss of renal function in patients with significant renovascular disease, or a pre-renal state.
- A number of drugs are also associated with the development or worsening of hypertension which may exacerbate renal disease (Box 10.3).
Determine the duration of drug use. Long-term use of certain analgesics (particularly phenacetin and combination analgesics containing caffeine and aspirin) and lithium has been associated with progressive renal impairment. Although now used less commonly in rheumatoid arthritis, prolonged use of gold salts and d-penicillamine may induce membranous nephropathy.
Steroids: Calcineurin inhibitors: Erythropoiesis-stimulating agents Sympathomimetic agents:
Patients may not regard ‘over-the-counter’ medication and herbal therapies as potentially harmful. It is therefore crucial to specifically ask about such therapies since they may contain potential nephrotoxins (for example aristolochic acid nephropathy caused by the ingestion of ‘Chinese herbs’).
Finally, some patients take medication surreptitiously. For example, laxatives or diuretics may be used as aids to weight loss, but can lead to severe potassium depletion or rebound oedema.
Dietary history
Enquiry into the dietary habits of patients can be informative.
- Excessive salt intake may result in significant hypertension and resistance to antihypertensive therapy. In patients with advanced CKD, significant volume overload and potentially life-threatening pulmonary oedema may be precipitated.
- Patients with renal stones tend to ingest large amounts of animal protein. This results in the increased urinary excretion of calcium, oxalate, and uric acid, and a low urine pH.
- Inadequate fluid intake may promote renal stone formation, particularly in those with an underlying metabolic predisposition to stone disease.
- Excessive alcohol intake leads to an increase in blood pressure and may result in poor concordance with therapy.
- A high intake of fruit juices and other acidic foods increases oxalate intake and may result in calcium oxalate precipitation in the kidney.
- The methylxanthine present in tea and coffee can induce polyuria in those with substantial intakes.
Social history
The socioeconomic and educational status of a patient can have an impact on the development of renal disease:
- The frequency of bacteriuria is much greater in pregnant women from the low socioeconomic group.
- Concordance with therapy is less good in hypertensive patients from the low socioeconomic group.
- Calcium stones form more frequently in men from higher socioeconomic groups, which probably relates to the increased protein intake associated with increasing affluence.
- Tobacco is a significant risk factor for the development and progression of renal disease. It contributes to the accelerated development of atherosclerosis in patients with CKD and is a risk factor for renovascular hypertension, accelerated hypertension, Goodpasture’s syndrome, and the development of diabetic nephropathy.
- Illicit drug use is associated with a number of renal complications including AKI due to rhabdomyolysis, bloodborne-virus-related renal disease, AA amyloidosis, vasculitis and proliferative glomerulonephritis.
Occupational history
The likelihood of developing renal disease is increased by a number of occupational factors.
- Working in a hot atmosphere results in increased insensible fluid loss, which increases the incidence of renal stone formation.
- The use of aniline dyes in the workplace increases the incidence of urothelial tumours.
- Exposure to inhaled hydrocarbons increases the incidence of Goodpasture’s disease.
- A number of infections can also be picked up in the workplace. Miners, sewage workers, and farm labourers are at an increased risk of leptospirosis. Hantavirus infection may arise in laboratory workers handling rodents, or in farmers working in endemic areas.
- Exposure to lead, particularly in a vaporized form (e.g. welding lead pipes), may result in lead nephropathy.
Family history
The exploration of the family history is important for the identification of hereditary renal diseases. It should also be appreciated that a number of other conditions without a clear inheritance pattern, such as systemic lupus erythematosus, IgA nephropathy and diabetic nephropathy, appear to have a genetic predisposition. A classification of inherited renal disease is shown in Table 10.3.
Ethnic and geographical factors
The ethnic background of a patient may impact on the risk of developing certain renal diseases. For example, in the UK:
- African Caribbeans with hypertension or diabetes mellitus: renal failure is more common than in white populations.
- South Asians and African Caribbeans: the incidence of end-stage renal disease is it at least three times higher than in Caucasians.
- White populations and from some parts of Asia: IgA nephropathy is more common than in black populations.
- Oriental, Hispanic and black populations: the incidence and severity of systemic lupus erythematosus is greater than in Caucasians.
- Arabs, Turks and Sephardic Mediterranean Jews (compared to Sephardic Jews from other regions, Ashkenazi Jews and Armenians): amyloidosis as a complication of familial Mediterranean fever occurs more often.
- Indians or Russians from high tuberculosis prevalence regions: the risk of developing tuberculosis during immunosuppressive therapy (for example after renal transplantation) is higher than in those from low prevalence areas.
PHYSICAL EXAMINATION
General examination
In patients with renal disease, the general examination can provide a wealth of diagnostic information.
Skin
- A decrease in skin turgor is a late sign of volume depletion.
- Uraemic patients frequently have dry and flaky skin with a yellowish-brown hue.
- Scratch marks or reddish-brown papules may indicate pruritus.
- Pallor is common in patients with CKD as a consequence of renal anaemia.
- Diffuse hyperpigmentation of sun-exposed areas may develop in patients with CKD.
- Subcutaneous bruising following minimal trauma is common and reflects disordered platelet activity and fragile cutaneous blood vessels. These lesions often result in discrete areas of hyperpigmentation due to haematin deposition (Fig. 10.2).
- Subcutaneous nodules caused by the deposition of calcium salts within the skin (calcinosis cutis) occur in patients with secondary and tertiary hyperparathyroidism.
- Cutaneous vasculitis can have many appearances including allergic-like exanthema, petechiae and purpura, and lesions with evidence of necrosis and subcutaneous haemorrhage (Figs 10.3 and 10.4).
- Small reddish-purple papules (angiokeratoma) located in the lower abdomen and groin area are suggestive of Fabry’s disease.
- An allergic exanthema may be associated with an allergic interstitial nephritis.
- In advanced renal failure, the accumulation and crystallization of urea and other nitrogenous waste products in sweat results in the deposition of a white crystalline material on the skin, particularly on the face (uraemic frost). This is now very rarely seen.
- Patients on long-term corticosteroids lose subcutaneous tissue and the skin becomes thin, fragile, and prone to low trauma bruising.
Nails
- Transverse ridges (Beau’s lines) indicate a serious preceding illness.
- Leuconychia (white nails, Fig. 10.5) can be seen in patients with nephrotic syndrome. If the nephrotic syndrome is transient or recurrent, transverse white bands may be seen (Muehrcke’s bands).
- Splinter haemorrhages or nail-fold infarcts are features of vasculitis or infective endocarditis.
- Nail dystrophy can be seen in the nail–patella syndrome and is usually associated with absent or hypoplastic patellae.
Face
- Thickening and rigidity of the facial skin with multiple telangiectasia suggests scleroderma/systemic sclerosis.
- A facial rash with a butterfly distribution may be associated with lupus erythematosus.
- Loss of the nasal cartilaginous septum leading to collapse of the nasal bridge is characteristic of Wegener’s granulomatosis.
- Loss of the buccal pad when accompanied by fat loss from the upper part of the body (partial lipodystrophy) is associated with mesangiocapillary glomerulonephritis type 2 (dense deposit disease).
- A moon face may be observed in patients treated with high-dose corticosteroids.
Eyes
- Perilimbal calcification in patients with long-standing uraemia.
- Subconjunctival haemorrhages in patients with vasculitis.
- Lenticonus in patients with Alport’s syndrome.
- Cataracts in patients who have had high-dose or prolonged corticosteroid use.
- The characteristic abnormalities of diabetic retinopathy, hypertensive retinopathy or vasculitis on fundoscopy
Chest
Examine the chest to identify:
- pleural effusions – which may arise from volume overload or the nephrotic syndrome
- lung crackles – which may represent volume overload or lung fibrosis
- pleural rubs – which may be caused by uraemia or inflammatory disease such as vasculitis.
Patients with metabolic acidosis may have a Kussmaul’s respiratory pattern (deep, sighing breaths).
Praecordium
- Examine the praecordium for the presence of murmurs and a pericardial rub. Carefully document murmurs since an evolving murmur may indicate a diagnosis of infective endocarditis with an associated immune complex-mediated glomerulonephritis.
- Flow murmurs are relatively common in patients with CKD as a consequence of renal anaemia or valvular sclerosis. Aortic, mitral and tricuspid incompetence can be found in patients with autosomal dominant polycystic kidney disease.
- A pericardial rub (pericarditis) can be a manifestation of uraemia (in which case it is an absolute indication to commence dialysis), infection or vasculitis.
Limbs
- Examine for the presence of oedema, which tends to settle in the legs under the effects of gravity. In bedridden patients oedema may be seen in the calves and thighs and also in the presacral area.
- Examine the vascular tree by palpating all pulses and auscultating for vascular bruits.
- Atherosclerotic peripheral vascular disease may be associated with renovascular disease and is also a common complication of CKD. In addition, the identification of peripheral vascular disease is relevant for future vascular access planning (for haemodialysis) and for assessing suitability for renal transplantation.
- Perform a neurological examination to exclude polyneuropathy that may be secondary to uraemia or diabetes mellitus. Polyneuropathy due to renal disease is symmetrical with predominance of the lower limbs. It may cause sensory loss, paraesthesia, dysaesthesia, itch and muscular cramps. A proximal myopathy may also complicate CKD.
- Examine the patellae for abnormalities which may suggest the presence of the nail–patella syndrome – small, poorly developed finger and toe nails and patellae.
Blood pressure
The accurate measurement of blood pressure is a vital part of the clinical evaluation of patients with renal disease. The procedure for measuring blood pressure is as follows.
- Sit the patient quietly for at least 5 minutes. They should be relaxed and not moving or speaking.
- Support the arm at heart level and ensure it is not constricted by tight clothing.
- Use an appropriately sized cuff: the bladder should enclose between 80 per cent and 100 per cent of the arm. If the cuff bladder is too small the blood pressure tends to be overestimated (by up to 30 mmHg in obese patients) whereas a cuff that is too large will underestimate the blood pressure (by 10–30 mmHg).
- When using a mercury manometer ensure that the mercury column is vertical and at the observer’s eye level.
- Estimate the systolic blood pressure by palpating the brachial artery, and inflating the cuff until pulsation disappears. Deflate the cuff and then reinflate to 30 mmHg above the estimated systolic pressure needed to occlude the brachial pulse.
- Place the stethoscope diaphragm over the brachial artery and deflate the cuff at a rate of 2–3mm/ second. Record the systolic (first Korotkoff sound) and diastolic (disappearance of sound) pressures to the nearest 2 mmHg. If the Korotkoff sounds do not disappear (e.g. patients with aortic valvular insufficiency or a high cardiac output) record phase IV (muffling of sound) as the diastolic blood pressure.
- Increasingly mercury manometers are being replaced by electronic manometers due to concerns over the toxicity of mercury. Ensure that these are validated devices, and that they are calibrated on a regular basis.
Examination of the kidneys and urinary tract
Direct examination of the kidneys is usually not possible so a careful general examination is necessary to identify any abnormalities suggesting underlying renal disease. Nonetheless, an examination of the abdomen can often be informative when significant pathology is present.
Clinical examination of the urinary tract follows the standard pattern of inspection, palpation, percussion and auscultation.
Inspection
On abdominal inspection in thin patients, it may occasionally be possible to see one or both kidneys, particularly if polycystic kidney disease is present. Obstruction of the bladder outflow tract may result in bladder distension which may be visible as a suprapubic mass.
Rarely, chronic urinary tract obstruction may cause such distension of the ureters that they become visible.
Palpation
Palpation of normal kidneys is usually only possible in thin patients. Palpation is best carried out with the patient in the recumbent position with the head supported on a pillow and the arms resting at the side of the body. To palpate the right kidney, place the left hand posteriorly in the loin and the right hand horizontally on the anterior abdominal wall to the right of the umbilicus. By pushing forward with the left hand and asking the patient to take a deep breath, the lower pole of the kidney may be palpable by pressing the right hand inwards and upwards.
The left kidney is not as readily palpable as the right. To palpate the left kidney, place the left hand posteriorly in the left loin, and the right hand on the anterior abdominal wall to the left of the umbilicus. If the kidney is palpable, estimate its size and shape. Normally the surface of the kidney is smooth and relatively hard, but in cystic disease an irregular surface may be appreciated. Note any tenderness on palpation.
Percussion
- In patients with acute pyelonephritis or acute glomerulonephritis, percussion of the renal region may cause severe pain (costovertebral tenderness).
- Pyelonephritis is usually accompanied by fever and dysuria, whereas haematuria, hypertension and oliguria suggest the presence of glomerulonephritis.
- Abdominal percussion may be of value if there is difficulty distinguishing between an enlarged kidney (resonant) and splenomegaly or hepatomegaly (dull).
Auscultation
Abdominal auscultation for vascular bruits is essential in patients with either hypertension or unexplained CKD (particularly if the urinalysis is negative and there is other evidence of atherosclerotic vascular disease). Place the stethoscope posteriorly in the loin, laterally in the flank, and anteriorly, and in each area listen for a bruit.
INVESTIGATIONS
Urinalysis
Urine examination is one of the basic diagnostic tests for patients with renal tract disease. Urine can be evaluated in a number of ways.
Urine dipstick testing
Urine dipstick testing is a widely available bedside test that can provide a wealth of diagnostic information. Dipsticks can detect blood, protein, glucose, ketones, nitrites and leucocyte esterase, and can measure the urine pH. The implications and limitations of dipstick testing are listed in Table 10.4. The key findings of relevance for renal disease are blood and protein. Assuming infection has been ruled out, their presence indicates either structural abnormality in the renal tract or glomerular disease, and therefore persistent abnormalities always require appropriate specialist evaluation. The urine dipsticks are essentially specific for albumin. They therefore do not detect other proteins that may be of relevance to renal disease (e.g. Bence Jones proteins – see below).
Urine microscopy
In certain circumstances urine microscopy can provide important diagnostic information. The important findings on urine microscopy are shown in Table 10.5.
Specific biochemical tests
In certain situations, undertake specific tests of the urine.
- Quantitative measurement of proteinuria: Until recently, this was performed on 24-hour urine collections. However, these are notoriously unreliable and therefore measurement of the urine albumin (or protein):creatinine ratio (ACR and PCR, respectively) on a spot urine sample is the currently preferred method. A PCR of less than 15 mg/ mmol is normal, and equates to less than 150 mg of proteinuria per day. A PCR of greater than 350 mg/ mmol represents nephrotic range proteinuria.
- Urine immunoelectrophoresis: This is undertaken to detect immunoglobulin light chains (Bence Jones protein) in patients in whom myeloma is suspected.
- Metabolic screen for stone disease: In patients who form recurrent renal stones, investigate for an underlying metabolic abnormality by measuring calcium, oxalate, urate and cystine excretion in 24-hour urine collections.
Blood
Blood tests are required to:
- assess renal excretory function
- identify the metabolic consequences of CKD
- identify immunological markers of systemic disease that may be associated with renal disease.
To assess renal excretory function, measure the serum creatinine concentration. From this, the estimated glomerular filtration rate (eGFR) can be calculated. The commonly used calculation includes the serum creatinine concentration, age, gender and a correction factor for black subjects. eGFR measurements can be used to categorize the severity of renal disease into CKD stages 1–5 (see Table 10.6).
To assess the complications of CKD, measure the serum phosphate, calcium, bicarbonate, potassium, parathyroid hormone, and haemoglobin. Additional serological tests that are useful in determining the cause of renal disease are shown in Table 10.7.

Imaging
Ultrasound scanning
This is the first-line imaging investigation for the majority of kidney or urinary tract diseases. It is non-invasive, requires no toxic contrast media, requires minimal patient preparation and avoids the use of ionizing radiation. It provides no functional information, however. The indications for ultrasound are to:
- assess the size and position of the kidneys
- look for evidence of cortical scarring
- demonstrate and characterize renal masses (solid and cystic)
- demonstrate bladder abnormalities
- identify renal tract obstruction
- demonstrate renal calcification and stone formation.
Intravenous urography
This was commonly used to look for structural abnormalities of the urinary tract and was particularly good for conditions involving the urothelium. Its use, however, has been largely superseded by computed tomography.
Computed tomography
Non-contrast enhanced computed tomography (CT) is the first line investigation for patients with suspected ureteral colic. It has a greater than 95 per cent sensitivity and specificity for stone disease. Contrast-enhanced CT is used to:
- characterize and stage urogenital cancer
- differentiate cystic from solid lesions
- demonstrate soft tissue abnormalities related to the renal tract
- identify collections associated with the renal tract G evaluate the renal tract after trauma
- identify renal artery stenosis and adrenal tumours in hypertensive patients.
Computed tomography involves a significant radiation dose and also the administration of contrast media which can be nephrotoxic, particularly in patients with impaired renal function.
Magnetic resonance imaging
This is increasingly being used to:
- characterize renal mass lesions
- stage bladder and prostate carcinoma
- assess the presence of tumour thrombus in renal tumours
- detect renal artery stenosis.
It avoids the need for ionizing radiation and the gadolinium-based contrast medium is non-nephrotoxic. However, gadolinium has been associated with a scleroderma-like skin condition (nephrogenic systemic fibrosis) in a small number of patients with significantly impaired renal function. Current guidance is to avoid gadolinium-containing contrast media, if possible, in patients with an eGFR of less than 30 mL/minute.
Renal biopsy
In some patients, examination of blood and urine and imaging investigations are unable to provide a specific diagnosis. Therefore in patients with evidence of glomerular disease (haematuria, proteinuria, impaired renal function) or renal failure of unknown cause, a renal biopsy may be necessary to determine the precise diagnosis. The indications for renal biopsy are shown in Box 10.4.
Although renal biopsy is a routine investigation for the evaluation of patients with renal disease, it is not without risk and therefore the decision to undertake a renal biopsy should be made by a specialist renal physician. Involve nephrologists early in the management of patients in whom intrinsic renal disease is suspected.
COMMON DIAGNOSES
Nephrotic syndrome
Nephrotic syndrome is the term given to the constellation of:
- heavy proteinuria >40 mg/m2/h or >50 mg/kg/ day; for a 70 kg man, this means more than 3.5 g/ day
- hypoalbuminaemia (<25 g/L)
- oedema
- hyperlipidaemia
- lipiduria.
Nephrotic syndrome is a potentially serious condition that is associated with a variety of other complications including hypercoagulability, negative nitrogen balance and infection. The major causes are listed in Table 10.8.
History
Nephrotic patients usually present with oedema which is often first noticed in a peri-orbital distribution after a period of recumbency. This distribution is characteristic of nephrotic syndrome, since patients with oedema due to heart failure or cirrhosis are often unable to lie flat because of pulmonary oedema or pressure on the diaphragm, respectively. As the oedema worsens, fluid accumulates around the ankles, sacrum and elbows in a gravity-dependent manner, and then becomes more generalized. Genital oedema may ensue and cause significant distress. A rapid onset of oedema suggests minimal change disease or focal segmental glomerulosclerosis (FSGS), rather than other causes in which the onset may be more insidious.
Nephrotic patients often describe frothing of the urine on micturition. Visible haematuria in nephrotic patients is rare, but when reported may indicate postinfectious glomerulonephritis, or the development of renal vein thrombosis.
Other key aspects of the history to explore include:
- medication (prescribed or over-the-counter)
- prior acute or chronic infection
- allergies
- features suggestive of a systemic disorder such as lupus erythematosus or diabetes mellitus.
In patients older than 50 years, membranous nephropathy may be associated with epithelial tumours, particularly of the lung, breast or large bowel, therefore enquire about symptoms of malignancy.
A positive family history may indicate Alport’s syndrome or an inherited form of focal segmental glomerulosclerosis.
Examination
Severe oedema may be associated with striae even in the absence of steroid therapy. Asymmetrical leg swelling may indicate deep venous thrombosis. Pleural effusion and ascites may be present. The liver may be painlessly enlarged, particularly in children. The jugular venous pressure is usually normal or low, and if elevated co-existent cardiac disease has to be suspected. Amyloidosis may account for both nephrotic syndrome and heart failure in this setting.
White bands (Muehrcke’s bands) may be identified on the nails; these correspond to periods of hypoalbuminaemia. Hyperlipidaemia associated with the nephrotic syndrome may result in the formation of xanthoma around the eyes or along tendons.
Investigations
- Urine dipstick will be strongly positive for protein and may also be positive for blood.
- The urine protein:creatinine ratio will be >350 mg/mmol.
- Urine microscopy may demonstrate oval fat bodies and fatty casts.
- Renal function may be normal or impaired.
- Serum albumin concentration will be reduced, usually to less than 25 g/L.
- Cholesterol is usually elevated, often substantially, with 25 per cent of patients having a total cholesterol >10 mmol/L.
- Appropriate immunological investigations include: serum immunoglobulins; antinuclear antibody; and complement components C3 and C4 (Box 10.4 and Table 10.8).
- In patients >40 years send serum and urine for protein electrophoresis.
The majority of patients with nephrotic syndrome will require a renal biopsy to determine the underlying cause. However, in patients with longstanding diabetes mellitus with evidence of retinopathy and peripheral neuropathy a diagnosis of diabetic nephropathy can be assumed.
A presentation with facial oedema usually indicates a diagnosis of nephrotic syndrome.
Nephritic syndrome/rapidly progressive glomerulonephritis
These syndromes result from glomerular inflammation and cause loss of excretory function, fluid retention, and haematuria. Rapidly progressive glomerulonephritis (RPGN) is a severe form of nephritic syndrome which leads to AKI, and may not be self-limiting. Common diseases presenting with the nephritic syndrome/RPGN together with associated features are listed in Table 10.9.
History
The classic nephritic syndrome presentation is:
- oliguria of rapid onset
- weight gain
- generalized oedema, which develops over a few days
- haematuria, which typically manifests as red/ brown urine (often described as smoky or cola coloured). Blood clots are never seen.
Other key aspects of the history to explore include:
- a history of prior infection (often pharyngitis or impetigo)
- constitutional symptoms (weight loss, fever, myalgia, arthralgia) which may be associated with vasculitis
- skin rashes which may accompany vasculitis and lupus erythematosus
- respiratory tract symptoms (nasal congestion and bleeding, sinus congestion/infection, deafness, haemoptysis) which are suggestive of Wegener’s granulomatosis
- uraemic symptoms.
Obtain a smoking history, and examine the occupational history for exposure to hydrocarbons.
Examination
Examine for volume overload:
- peripheral oedema is usual, but less severe than would be seen in the nephrotic syndrome
- hypertension is invariable
- pulmonary oedema may develop (combination of hypertension and volume overload)
- elevation of the jugular venous pressure is common.
Other key aspects of the examination include:
- skin rashes
- splinter haemorrhages and nail-fold infarcts
- ENT disease (nasal mucosal inflammation, sinus tenderness, middle ear effusion)
- eyes for subconjunctival haemorrhage and evidence of retinal vasculitis
- lung crackles, pleural rubs
- limbs for neurological deficit which may result from nerve injury due to vasculitis.
Investigations
- Urine examination will demonstrate haematuria and proteinuria (usually subnephrotic).
- Urine microscopy may demonstrate red cell casts. G Blood tests will show reduced renal function, often an elevated C-reactive protein, and anaemia. G Additional serological investigations are listed in Tables 10.7 and 10.9.
- A renal biopsy is usually required in patients with RPGN unless an associated infection is identified, in which case a postinfectious glomerulonephritis can be assumed.
A patient with acute kidney injury who has haematuria and proteinuria on urine dipstick testing should be considered to have rapidly progressive glomerulonephritis and specialist nephrology input should be sought immediately.
IgA nephropathy/Henoch–Schönlein purpura
IgA nephropathy, characterized by the mesangial deposition of IgA, is the commonest glomerulonephritis in the Western world. The typical clinical presentations are:
- visible haematuria (30–40 per cent of patients) usually 12 to 72 hours after the development of a mucosal infection (usually pharyngitis) or after exercise
- asymptomatic (30–40 per cent of patients) identified on incidental urine testing (haematuria ± proteinuria)
- nephrotic syndrome (5 per cent of patients)
- RPGN (5 per cent of patients)
- severe hypertension (occasional patient)
- loin pain (unilateral or bilateral) (occasional patient).
Henoch–Schönlein purpura is a related disease with the same glomerular abnormalities as IgA nephropathy, but with evidence of systemic vasculitis. This typically presents with:
- a palpable purpuric rash in an extensor surface distribution with sparing of the trunk and face (Fig. 10.6)
- polyarthralgia
- abdominal pain, which in some cases may be severe (presenting as an acute abdomen) and associated with bloody diarrhoea.
There are no signs that are characteristic of IgA nephropathy, and in Henoch–Schönlein purpura the only specific sign is the characteristic purpuric rash.
There are no specific laboratory investigations that are diagnostic of either IgA nephropathy or Henoch–Schönlein purpura, although up to 30 per cent of patients will have elevated serum IgA levels. A renal biopsy is essential to make a diagnosis of IgA nephropathy.
In children, the constellation of haematuria/proteinuria, purpuric rash, arthralgia and abdominal pain is usually sufficient to make a clinical diagnosis of Henoch-Schönlein purpura without the need for renal biopsy.
Diabetic nephropathy
Diabetic renal disease is the commonest cause of end-stage renal disease in the Western world. It accounts for 20–50 per cent of new patients with end-stage renal disease.
Diabetic nephropathy typically presents with a constellation of:
- nephrotic range proteinuria
- hypertension
- progressive renal failure
- overt nephrotic syndrome in some patients (diabetic nephropathy is the commonest cause of nephrotic syndrome).
Key aspects of the history that are useful in making a diagnosis of diabetic nephropathy include:
- diabetes mellitus of long duration (at least 10 years)
- poor glycaemic control
- inadequately controlled hypertension
- microvascular disease in other organ systems, particularly diabetic retinopathy and peripheral neuropathy. Note that some patients with type 2 diabetes can have diabetic nephropathy without retinopathy
- cigarette smoking, ethnicity (Indo-Asians, African Americans), family history of diabetic nephropathy, and socioeconomic factors (poverty) are all risk factors favouring the development of diabetic nephropathy.
Key aspects of the examination are:
- funduscopy for diabetic retinopathy and limb examination for evidence of peripheral neuropathy
- volume state examination for evidence of the nephrotic syndrome
- blood pressure measurement
- examination of the vascular tree for evidence of atherosclerotic vascular disease, including renal bruits.
Investigations
- Proteinuria on urine dipstick.
- Urine protein:creatinine ratio >350 mg/mmol.
- Blood tests will demonstrate impaired renal function, elevated HbA1c, elevated triglycerides.
There are no other specific serological abnormalities diagnostic of diabetic nephropathy. Mostly a diagnosis of diabetic nephropathy can be made confidently on clinical grounds and renal biopsy is reserved for cases with atypical features (haematuria, short duration of diabetes, rapid decline in renal function, rapid onset of nephrosis, abnormal immunological investigations).
Myeloma kidney
Abnormal immunoglobulin proteins produced by malignant plasma cells can produce a variety of renal abnormalities. The commonest renal lesion is myeloma cast nephropathy (myeloma kidney). Patients may present with:
- the systemic effects of the plasma cell malignancy: weight loss, malaise, anaemia (fatigue and exertional breathlessness), bone pain, symptoms of hypercalcaemia (polyuria, thirst, abdominal pain, bone pain), and recurrent infection
- renal dysfunction in patients already known to have myeloma
- isolated renal failure without any associated features. Myeloma kidney is diagnosed on renal biopsy or from the blood and urine investigations undertaken in patients with unexplained renal failure.
Physical examination is usually unhelpful in myeloma patients. Pallor may be present. Bone deposits may cause spine tenderness or deformity. Occasionally mass lesions (plasmacytoma) may be identified.
The diagnosis depends on:
- a serum or urine monoclonal paraprotein with suppression of other immunoglobulin classes (serum and urine protein electrophoresis)
- an excess of plasma cells in the bone marrow (bone marrow biopsy)
- the presence of lytic bone lesions on plain radiography (skeletal survey).
Additional investigations required include full blood count to assess marrow function, and serum biochemistry to assess renal function and to identify hypercalcaemia.
If the urine dipstick is negative when other tests (e.g. urine protein:creatinine ratio) indicate significant proteinuria, consider myeloma (dipsticks are albumin-specific and do not detect immunoglobulin light chains).
Autosomal dominant polycystic kidney disease
Autosomal dominant polycystic kidney disease (ADPKD) is a common genetic cause of renal disease with a prevalence of 1:400–1:1000 live births. It accounts for 10 per cent of patients with end-stage renal disease. Patients are often asymptomatic and present with either hypertension, an incidental finding of renal cysts on abdominal ultrasound, or a family history of renal disease.
When specifically questioned, the symptoms at presentation include:
- flank, back or abdominal pain in 60 per cent
- hypertension in 60 per cent
- haematuria in 75 per cent (30–50 per cent visible, 25 per cent invisible)
- a history of urinary tract infection
- uraemic symptoms in a minority. This is most likely with late presentation in the fourth to sixth decades of life.
A family history of renal failure or cystic renal disease is present in up to 90 per cent of patients. Ten per cent of cases appear to be due to new mutations in the PKD genes. Ten per cent of patients will have a family history of intracranial bleeds due to berry aneurysms.
The examination findings include:
- bilateral irregular flank masses in the abdomen
- nodular hepatomegaly (from liver cysts) (75 percent of patients by the seventh decade)
- aortic regurgitation, mitral valve prolapse, or tricuspid regurgitation may be detected due to associated cardiac valve defects
- inguinal and umbilical hernias may also be present.
Relevant investigations include:
- serum chemistry profile – to assess renal function G full blood count – although anaemia in ADPKD is less common than in other forms of CKD
- urinalysis – this will demonstrate invisible haematuria in 25 per cent and proteinuria in 18 per cent
- ultrasonography – this is the most appropriate imaging modality to identify the renal cysts
- cerebral magnetic resonance angiography – to look for berry aneurysms; it should be reserved for patients with a family history of intracerebral bleeding (10 per cent of families).
Alport’s syndrome
Alport’s syndrome is an inherited syndrome of haematuria, progressive renal impairment, sensorineural deafness, and eye abnormalities. It has a prevalence of 1:5000 live births, and accounts for 2–3 per cent of patients with end-stage renal disease. An X-linked inheritance pattern is present in 80 per cent of cases, with autosomal recessive and autosomal dominant forms accounting for 15 per cent and 5 per cent of cases, respectively. All forms are caused by mutations in type IV collagen genes (usually the COL4A5 gene). The presentation can be variable.
- Some patients present with intermittent visible haematuria, often in association with respiratory tract infection.
- Nephrotic syndrome develops in 40 per cent of cases in early adulthood.
- End-stage renal disease and therefore uraemic symptoms usually develop during the third decade.
- Overt deafness becomes apparent during the second decade of life.
On examination, the key abnormalities are:
- hypertension – which is common once renal dysfunction has developed
- anterior lenticonus on formal eye examination – found in 15 per cent of patients, this correlates with the development of progressive renal dysfunction
- asymptomatic perimacular granulations on fundoscopy – found in 30 per cent of patients
- reduced hearing acuity – may be noticeable on bedside testing during the second decade.
Relevant investigations include:
- urinalysis demonstrates invisible haematuria from infancy
- progressive proteinuria develops from the second decade of life
- urea and creatinine become elevated as renal function declines and end-stage renal disease usually develops during the third decade
- renal biopsy is usually required for diagnosis, although audiometry and genetic testing may provide additional information.
Renal stone disease
Renal stone disease has a prevalence of up to 10 per cent in industrialized nations and is higher in some areas such as the Middle East. The modes of presentation are:
- pain – a stone passing through the renal pelvis or ureter induces severe colicky loin pain that radiates to the groin, testis, or labia (ureteral colic)
- urinary symptoms – stones impacted at the vesicoureteric junction may cause urinary frequency and pain in the penile tip or clitoris
- visible haematuria – this may occur during episodes of ureteral colic, or may occur in isolation
- recurrent urinary tract infection
- incidental finding – stones that are confined to the renal pelvis are often asymptomatic and are usually identified in imaging investigations undertaken for other reasons.
Bladder stones may cause haematuria, symptoms due to recurrent urinary sepsis, or frequency and urgency of micturition (stranguary) that can be very distressing. Seek out risk factors for stone disease (Box 10.5).
General risk factors: Environmental factors: Drug-related factors:
Examination of a patient with ureteral colic will demonstrate an agitated patient in obvious distress. The skin will be pale and clammy, although there is usually no haemodynamic compromise. Abdominal examination usually demonstrates no abnormality apart from mild loin tenderness. Fever and haemodynamic compromise in a patient with stone disease raises the possibility of sepsis from an obstructed kidney (pyonephrosis). Urgent drainage of the affected kidney after resuscitation and appropriate antibiotic treatment is required.
Appropriate investigations include:
- dipstick urinalysis – which will usually demon-strate invisible haematuria
- serum chemistry – for renal function, calcium and uric acid
- non-contrast enhanced spiral CT – currently the preferred option for identifying renal and ureteral stones.
In recurrent stone formers perform a metabolic screen (24-hour urine for calcium, phosphate, oxalate, uric acid, sodium, citrate, and pH) to determine whether there is an underlying metabolic abnormality that requires treatment to prevent further stone formation.
Fever, hypotension and loin tenderness in a patient with stone disease suggests the presence of an infected, obstructed kidney (pyonephrosis). Once resuscitated, urgent imaging and drainage of the kidney is required to prevent irreversible loss of renal function and potentially death.
Urothelial malignancy
The cardinal feature of transitional cell carcinoma of the urinary tract is painless haematuria. This may be visible or invisible. Any patient above the age of 40 years with haematuria requires urological evaluation as a priority.
Bladder tumours may cause irritative symptoms of frequency, urgency, nocturia, and dysuria. Loin pain may indicate the presence of ureteral obstruction. Weight loss and other symptoms of systemic malignancy develop late in the course of disease.
In the history, seek evidence of risk factors for urothelial malignancy. These include:
- cigarette smoking
- occupational exposure to aromatic amines and polyaromatic hydrocarbons
- previous exposure of the bladder to ionizing radiation (for example radiotherapy for cervical or prostate carcinoma)
- previous treatment with cyclophosphamide or phenacetin
- prolonged immunosuppression (for example in organ transplant recipients)
- chronic cystitis, bladder calculi, long-term indwelling catheters, and schistosomiasis all predispose to bladder squamous cell carcinoma.
On examination, signs of urothelial malignancy are uncommon. Advanced disease may be associated with cachexia, the presence of a pelvic mass, and irregular hepatomegaly.
Appropriate investigations include:
- urine cytology
- renal tract imaging with ultrasonography, contrast-enhanced CT or MRI
- cystoscopy and ureteroscopy.
Any patient above the age of 40 years with haematuria (visible or invisible) requires urgent evaluation by a urologist to look for malignant disease of the urinary tract.
Urinary tract infection
Urinary tract infection (UTI) is a very common medical problem with 50–60 per cent of women having at least one UTI during their lifetime. The risk in men is much lower at 5 per 10000 per year. The classic symptoms are frequency and urgency of micturition, dysuria, and/or suprapubic pain.
Fever is not a feature of lower UTI. Infection of the upper urinary tract is suggested by acute onset flank pain, nausea, vomiting, myalgia and fever. Lower tract symptoms may or may not be present.
Enquire about factors that predispose to UTI, including:
- pregnancy
- recent urinary tract instrumentation
- diabetes mellitus
- immunosuppression
- underlying disease that may predispose to urinary tract obstruction (stones, renal cysts, urinary tract malignancy, urinary diversion procedures e.g. ileal conduit)
- a functionally abnormal urinary tract (e.g. vesico-ureteric reflux, neurogenic bladder)
- renal failure
- renal transplantation
- prostatitis-related infection.
In addition, determine the relationship of UTI symptoms to sexual intercourse and the use of spermicidal agents.
On examination look for fever and assess the circulation to identify evidence of upper urinary tract infection and septic shock. Examine the abdomen. Lower urinary tract infection may be associated with suprapubic tenderness alone. The presence of significant loin tenderness suggests pyelonephritis. In men with UTI, perform a digital rectal exam; prostate tenderness suggests the presence of prostatitis.
The key investigations are:
- dipstick urinalysis – the presence of blood, protein, nitrites and leucocyte esterase strongly suggests UTI
- culture of a clean-catch urine specimen – a bacterial count of >105 colony-forming units (CFU)/mL is considered significant bacteriuria.
In uncomplicated UTI, no further investigations are necessary. Recurrent or upper tract UTI requires further investigation with imaging (ultrasonography, intravenous urography or CT) to look for evidence of structural or functional urinary tract disease.
SUMMARY
Key features of the history
Presenting symptoms:
- disorders of micturition:
- frequency of micturition
- poor urine stream
- dysuria
- disorders of urine volume:
- polyuria
- oliguria
- anuria
- disorders of urine composition:
- haematuria
- proteinuria
- pneumaturia
- miscellaneous (e.g. solid material in urine)
- pain
- oedema
- uraemic symptoms (Box 10.2)
- symptoms of systemic disease – infective, inflammatory or malignant disease.
The rest of the history:
- past medical history (Table 10.2)
- gynaecological and obstetric history – menstrual, contraceptive, and pregnancy history
- drug history – including all prescribed, over-the counter, and herbal remedies
- dietary history – sodium and protein intake
- social history – smoking history, alcohol intake, and socioeconomic status
- occupational history – identify risk of exposure to nephrotoxins and infectious agents
- family history – of renal disease and diseases associated with renal disease
- ethnic and geographical background.
Key features of the examination:
- examination of the kidneys and urinary tract
- palpation of the kidneys and bladder
- auscultation for vascular bruits
- blood pressure measurement
- general examination
- face – thickened skin, rash, loss of subcutaneous fat, moon face
- eyes – perilimbal calcification, subconjunctival haemorrhage, lenticonus, retinopathy
- skin – scratch marks, rashes, pigmentation, skin turgor
- nails – dystrophy, leuconychia, Beau’s lines, splinter haemorrhages
- praecordium – pericardial rub and cardiac murmurs
- chest – lung crackles, pleural effusions, pleural rubs, respiratory pattern
limbs – oedema, peripheral neuropathy, peripheral pulses and vascular bruits.
Attribution
Creative Commons Attribution 4.0 International License
This part contains content from OpenStax College, Anatomy and Physiology. OpenStax CNX. Download for free at http://cnx.org/contents/14fb4ad7-39a1-4eee-ab6e-3ef2482e3e22@15.1.
This part contains content reprinted by permission of the publisher, CRC Press, an imprint of Taylor & Francis Group, an Informa Business https://www.routledge.com/















